- Creating Molecules -
Development of PET molecular probes applicable to animal and human PET studies by pursuing the potentials of chemical biology and advanced synthetic organic chemistry as the axis of molecular science. With the objective of creating highly-functionalized molecular probes for facilitating PET molecular imaging science, we seek to develop the future medicinal chemistry and explore the new methodology in in vivo molecular science with the view of general versatility.

1) Development of new chemical methodologies and key concepts in molecular science for the promotion of PET molecular imaging science
Focusing on the potentials of PET imaging technology applicable to small animals and human clinical studies, we are engaged in developing the new labeling methodology of disease-associated proteins and the highly-efficient synthetic methodology, necessary for advancing medicinal chemistry for molecular probe development, and furthermore realizing rapid chemical reactions for introducing
11C and
18F as short-lived positron emission nuclides into carbon frameworks of bioactive organic compounds with the aim of creating PET molecular probes with chemical and metabolic stability at radiolabeling positions.
2) Development of disease-associated PET molecular probes
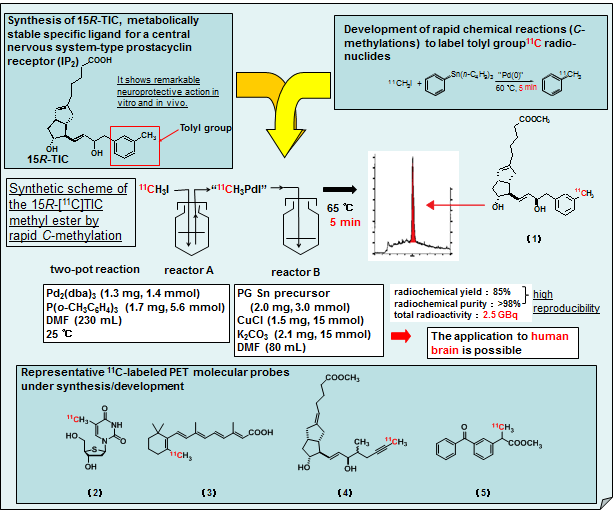
For demonstrating our methodologies, we are evolving the design and synthesis of PET molecular probes and exploring key compounds associated with new biological functions or important diseases such as cancers. Some of our present research activities are exemplified by development of 4 types of Pd
0-mediated rapid
C-[
11C]methylations on arene, alkene, alkyne, and alkane, realization of
11C-labeled 15
R-TIC methyl ester as a prostacyclin receptor ligand with specific neuroprotective effect for central nerves system, development of highly-efficient
11C-labeling of thymidine analogues for canner imaging, synthesis of
11C-labeled retinoic acids with anticancer effects, synthesis of
11C-labeled iloprost methyl ester with potent vasodilatory effects, and development of other rapid
C-[
11C]methylations via enolate intermediates representative by realization of
11C-labeled Ketoprofen methyl ester.

[Contact information for research-related enquiries]
Masaaki Suzuki TEL 078-304-7130 Mail
[Contact information for non-research-related enquiries]
Yasuko Kawai TEL 078-304-7130 Mail
![[Molecular Imaging Medicinal Chemistry Laboratory] Team Leader:Masaaki Suzuki](../image/laboratory/lob01_titleimage.jpg)



